What are GRPs?
grps
GRPs
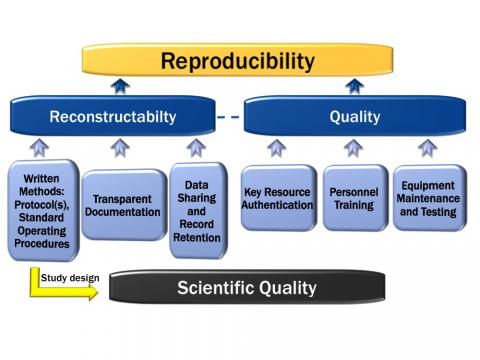
It is the center’s mission to support researchers through the development and support of Good Research Practices (GRPs). GRPs embody study planning, conduct, and reporting principles that share the intent of federal regulations (GLPs, see below).
Good Research Practices (GRPs) support:
- Meeting updated NIH/NSF and journal requirements for conducting and reporting research1-7
- Increased confidence in decisions based on reliable data
- Accessing industry partnerships more readily, and earlier

- 1 Collins, F. S. & Tabak, L. A. Policy: NIH plans to enhance reproducibility. Nature 505, 612-613 (2014).
- 2 Principles and Guidelines for Reporting Preclinical Research
- 3 Enhancing Reproducibility through Rigor and Transparency
- 4 McNutt, M. Journals unite for reproducibility. Science 346, 679 (2014)
- 5 Editorial. Journals unite for reproducibility. Nature 515, 7, doi:10.1038/515007a (2014)
- 6 Landis, S. C. et al. A call for transparent reporting to optimize the predictive value of preclinical research. Nature 490, 187-191, doi:10.1038/nature11556 (2012)
- 7 NSF Reproducibility Framework. (2014) (pdf)
More about GRPs
Written Methods: Protocol(s), SOP(s)
Purpose: To support the science by preserving the ‘memory’ of the experimental methods in time – if it was documented it can be fine-tuned. Documentation of the methods also supports NIH/NSF and journal requirements for conducting and reporting research1-6. Deviations from the written methods (protocol and/or SOP(s)) should be documented in order to ensure they are accurately described in reports.
- Study Protocol – approved and written methods that describe experimental design details and procedures that are specific to individual study(s). For example, the intervals of specimen collection, specific analyses to be performed, final disposition of study materials, etc.
- NIH has announced revised instructions to applicants and review criteria for funding proposals that include the following as part of the study protocol/plan.
- Scientific Premise: “… NIH expects applicants to describe the general strengths and weaknesses of the prior research…”
- Rigorous Experimental Design1,3: “NIH expects applicants to describe how they will achieve robust and unbiased results when describing the experimental design and proposed methods. Robust results are obtained using methods designed to avoid bias and can be reproduced under well-controlled and reported experimental conditions.”
- Consideration of Sex and Other Relevant Biological Variables: “NIH expects that sex as a biological variable will be factored into research designs, analyses, and reporting in vertebrate animal and human studies…Similarly, investigators should consider other biological variables, as appropriate, in the design and analyses of their proposed studies.”
- Authentication of Key Biological and/or Chemical Resources: “NIH expects that key biological and/or chemical resources will be regularly authenticated to ensure their identity and validity for use in the proposed studies.” For example, cell lines, specialty chemicals, antibodies and other biologics are considered key biological/chemical resources.
See also, New Grant Guidelines (NIH) (pdf), and Rigor and Reproducibility in NIH Applications Resource Chart (pdf). - Similar concerns regarding reproducibility and activity related to addressing these issues are underway through NSF – “The development of the NSF framework builds on considerable discipline-specific activity and infrastructure across elements of scientific research including: scientific milieu and question formulation; data collection, sharing, and curation; instrumentation, models, and, interpretation of findings; and research outputs…’6.
- NIH has announced revised instructions to applicants and review criteria for funding proposals that include the following as part of the study protocol/plan.
- Standard Operating Procedures (SOPs) – written methods used for training and reference by laboratory personnel that describe routine procedures in adequate detail to execute procedures consistently. Recommended SOPs include, but are not limited to:
- Equipment operations/maintenance/calibration (also see below, Equipment Standardization)
- Reagents and Solutions: Handling/labeling/disposal, etc.
- Data collection e.g. specimen/sample collection, processing, storage, etc.
- Other routine laboratory procedures e.g. animal dosing
- Data and record retention/archiving processes
- Training requirements
See Training and Resources for example SOP(s).
- 1Landis, S. C., Amara, S. G., Asadullah, K., Austin, C. P., Blumenstein, R., Bradley, E. W., . . . Silberberg, S. D. (2012). A call for transparent reporting to optimize the predictive value of preclinical research. Nature, 490(7419), 187-191. doi: 10.1038/nature11556
- 2Principles and Guidelines for Reporting Preclinical Research
- 3Enhancing Reproducibility through Rigor and Transparency
- 4 McNutt, M. Journals unite for reproducibility. Science 346, 679 (2014)
- 5Editorial. Journals unite for reproducibility. Nature 515, 7, doi:10.1038/515007a (2014)
- 6 NSF Reproducibility Framework. (2014) (pdf)
Transparent Documentation
Purpose: To preserve the ‘memory’ of the experiment in time
Good documentation supports full study reconstructability, aids in analysis, writing reports and fine-tuning methods.
Key elements of transparent documentation:
- Traceable – Who, When, Why, How
- Legible
- Complete and Contemporaneous – information is recorded with timeliness and is inclusive of all pertinent information
- Original – the first record of the information and/or the source information is retained
- Accurate - symbols and shorthand used in data recording are standardized and used consistently to avoid misinterpretation
- Durable -data are recorded in indelible ink to ensure integrity will be maintained over time, electronic data are treated with analogous considerations and maintained in a format that can be accessed over time. See also Data Sharing and Record Retention
Adapted from:
- Bargaje, C. (2011). Good documentation practice in clinical research. Perspect Clin Res, 2(2), 59-63. doi: 10.4103/2229-3485.80368
- Bhattacharya, M. (2014). Good Documentation Practice (Gap): Coordinate Regulatory Requirements in Pharmaceutical Manufacturing Industry. Journal of Pharmacology and Biological Sciences. 9(5), 24-37 (pdf)
Data Sharing and Record Retention
Purpose: To ensure accurate records are maintained and are readily accessible as per NIH/NSF and journal principles and guidelines for reporting preclinical research1,2
It is recommended that data and reports are maintained in a durable format using a consistent filing and retrieval system that ensures records can be readily provided if requested during consideration for publication and upon reasonable request upon publication as per ‘data and material sharing’ guidelines1 and expected policy changes requiring ‘on-line access to methods, protocols, original data, data reductions and analysis protocols'2.
Key Resource Authentication
Purpose: To maintain experimental consistency by ensuring key reagents and/or biologics are adequately validated.
It is recommended that key reagents (e.g., cell lines, specialty chemicals, antibodies and other biologics) are validated at intervals adequate to ensure the quality of the data. Methods used to validate these reagents should be documented and any data (generated and/or provided by supplier) are maintained with the study files as per data sharing and record retention guidelines.
See ‘Authentication of Key Biological and/or Chemical Resources’ under Written Methods.
Personnel Training
Purpose:
- Safety
- To ensure methods are executed consistently.
Differences in technique among personnel may be a source of unintentional method deviation(s). Training - in combination with comprehensive documentation of methods - can affect study reproducibility. It is recommended that training methods, frequency etc. are individualized at the discretion of laboratory managers and that training activities are documented.
Equipment Maintenance and Testing
Purpose: To ensure equipment are functioning adequately and consistently
Reliability of data is dependent on what can be consistently measured. The following are recommended guidelines for standardizing laboratory equipment:
- Equipment are operated and maintained according to the manufacturer’s recommendations. Exceptions are possible with justification.
- Written methods describe operation processes to facilitate training, consistent operation and data quality.
- Routine maintenance and calibration program procedures are also described in written methods for pertinent equipment (e.g., balances/scales, thermometers, incubators, volumetric pipettes, freezers/refrigerators etc.). These methods should describe, at a minimum, testing procedures and intervals, pass/fail limits, and actions to restore equipment in the case of malfunction.
- Equipment that are found defective or not maintained (e.g., for parts only) are labeled appropriately.
- Equipment used are identified in the study data e.g., if the data is questioned, the equipment involved in data collection can be identified and any records required to verify functionality of the equipment are traceable.
glps
GLPs
The GRP Resource Center has experience implementing Good Laboratory Practices (GLPs) - federal regulations that describe the minimum requirements for conducting non-clinical studies that support or are intended to support research or marketing permits for products regulated by the Food and Drug Administration (FDA) or Environmental Protection Agency (EPA).
The primary intent of the GLPs is to ensure data quality, integrity and reproducibility. To support this intent, the GLPs direct several conditions under which studies are initiated, planned, performed, monitored, recorded and reported. GLP elements are core components for conducting reproducible research that can be adapted to establish guidelines for Good Research Practices (GRPs).
- 1 CFR - Code of Federal Regulations Title 21. Part 58 Good Laboratory Practice for Nonclinical Laboratory Studies
- 2 CFR - Code of Federal Regulations Title 40, Parts 160 and 792 Good Laboratory Practice Standards