Sponsor-Investigator (GCP) CITI Training FAQs
1. Who must complete Sponsor-Investigator (GCP) training on CITI?
- IRB policy requires mandatory training for investigators who are also serving as sponsors, (e.g., holding an Investigational New Drug (IND), Investigational Device Exemption (IDE), or abbreviated IDE), for an FDA-regulated clinical investigation. A Sponsor-Investigator must complete the applicable drug or device Good Clinical Practice training on CITI before final IRB approval is granted. The ReGARDD Training Modules on IND Sponsor-Investigator Responsibilities [HTML] may be substituted for the CITI GCP course by providing completion documentation to ORI (HSPTrainingSupport@uky.edu).
- If the Sponsor-Investigator has documentation of completion of equivalent training, email course completion documentation to HSPTrainingSupport@uky.edu.
- Sponsor-Investigators may also choose to require completion of the training by staff who will be involved in the conduct of the study. This is encouraged for sub-investigators to whom any sponsor responsibilities will be delegated.
2. How do I complete the CITI training specifically required for Sponsor-Investigators of FDA regulated clinical research?
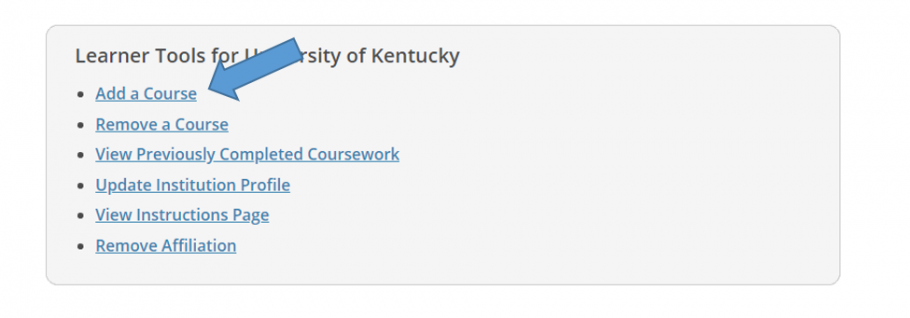
- You may enroll initially from the Courses page after logging in to your account. Scroll to the bottom of your Courses page then click "Add a Course" under Learner Tools for University of Kentucky.
-
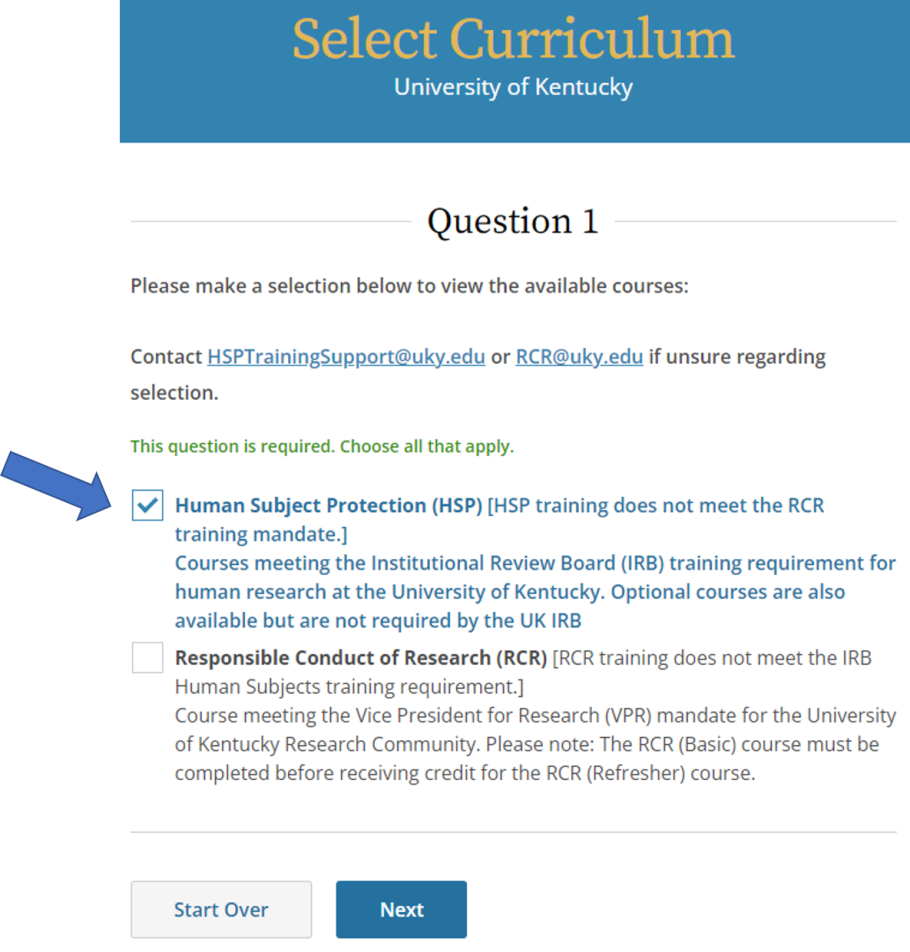
- Select Human Subject Protection (HSP), then click Next.
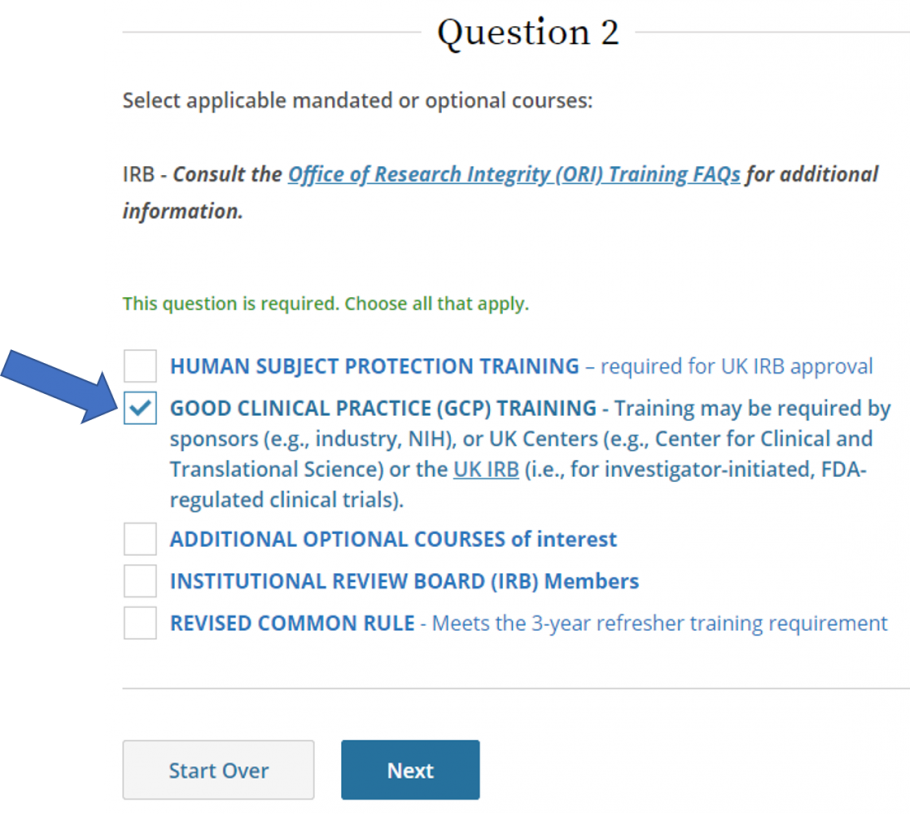
- Select Good Clinical Practice (GCP) training, then click Next.
-
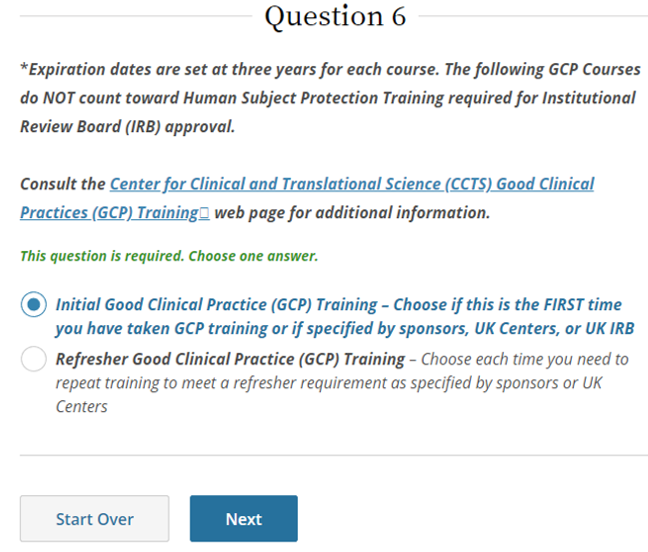
- Select Initial or Refresher, then click Next.
-
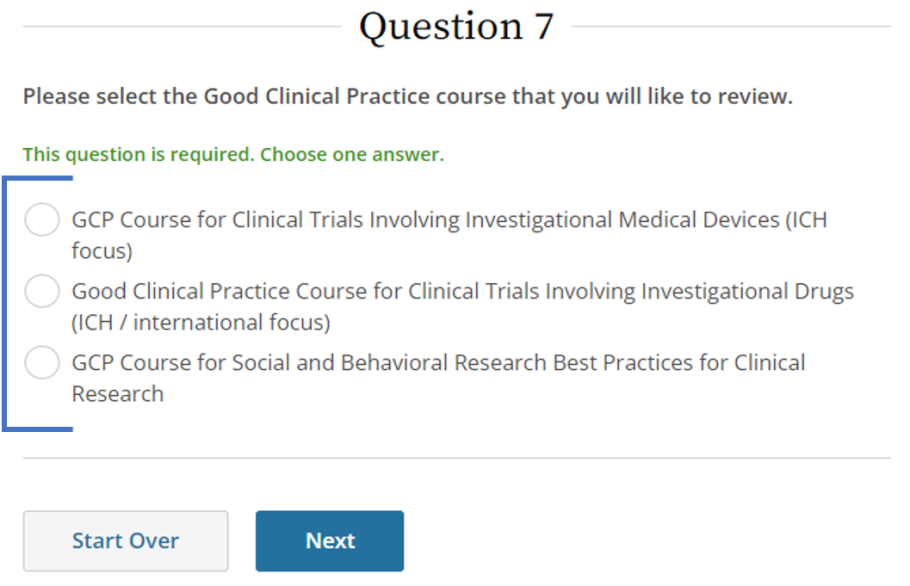
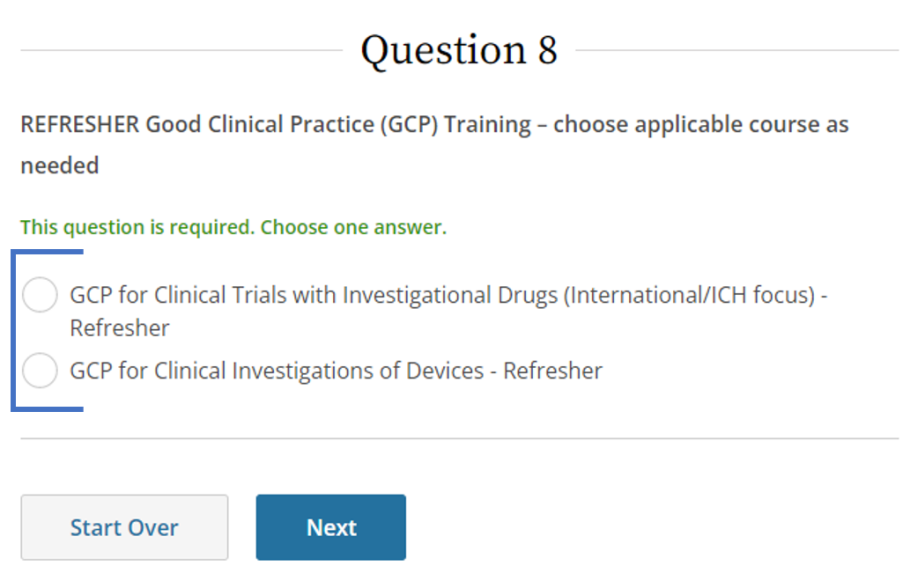
- Select applicable course, then click Next.
- Initial training:
-
- Or Refresher training:
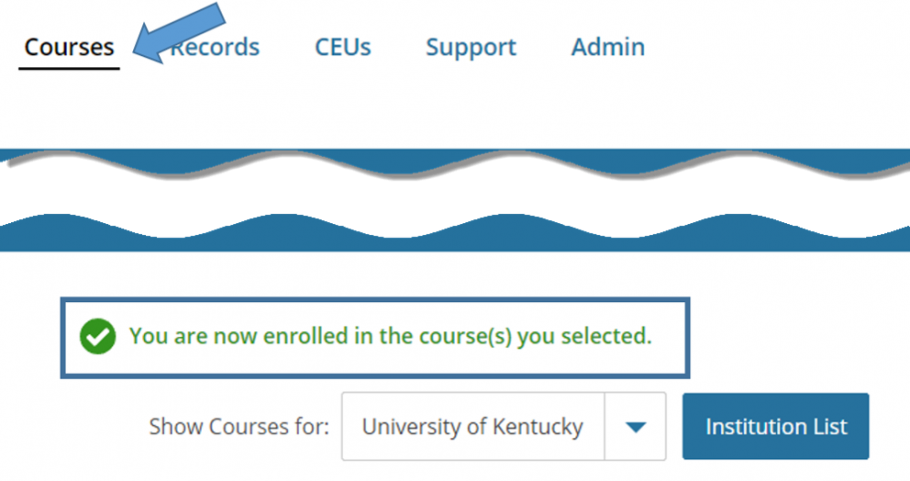
- You are directed back to your Courses page where it states you are now enrolled.
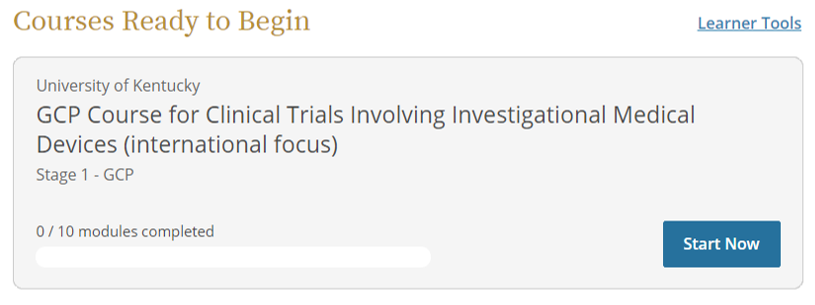
- Scroll down to "Courses Ready to Begin" and click Start Now to complete the Initial HSP training.
-